Challenge
A pharmaceutical company faced high variability in batch quality for an API process, resulting in a high risk of batch failure. This variability persisted despite good control over critical process parameters (CPPs) identified during research and development. The company needed to identify the root cause of the variability and implement corrective actions to improve process consistency and product quality.
Approach
The company adopted a data-driven approach to analyze the process and identify the source of variability. Data was extracted from various sources, including paper-based batch manufacturing records (BMRs), analytical reports, and SCADA sheets. This data was then digitized, resulting in approximately 8,000 pages of data and 40,000-50,000 data points.
Solution
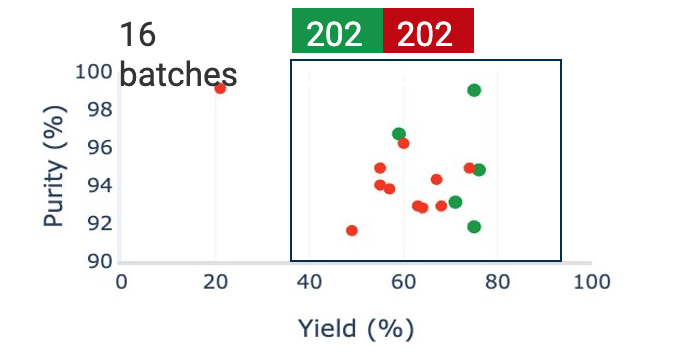
Automated statistical and correlation analyses were performed on approximately 2,000 process parameters, material attributes, quality attributes, and analytical parameters. This analysis revealed a strong correlation between yield and purity with batch size and the duration of the catalyst vessel rinsing step after catalyst addition.
Further analysis of approximately 700 parameters for each batch confirmed these correlations. The root cause of the variability was identified as poor control over the raw material (RM) heating profile, leading to variations in effective reaction time. Additionally, the rinse time of catalyst preparation was not controlled, causing further variations in total reaction time.
Results
Based on these findings, significant changes were made to the batch production record (BPR) procedures to improve control over the RM heating profile and catalyst preparation rinse time. These changes resulted in improved control over the process and a reduction in batch variability.
The time taken for analysis was 4 hours, and report preparation took 3 days.
Key Takeaways
- A data-driven approach can be highly effective in identifying and addressing root causes of variability in manufacturing processes.
- Automated data analysis tools can help to quickly analyze large datasets and identify critical correlations.
- Extracting data from various sources can provide a more complete picture of the process and enable a comprehensive analysis.
- Significant improvements in quality and yield can be achieved through process optimization based on data-driven insights.
Customer Details
Customer Product: New Small Molecule Drug API
Process Focus: Scale-up of Small Molecule Step
Customer Type: Leading CRDMO
Geography: USA and India
Nuron Products Used
Solutions: i-Extract, Data-to-Decision (API), i-Predict, i-Validate
Implementation: Automation of live OOT, OOS, and predictive decision-making.